|
8/17/2023 0 Comments Immune repertoire and disease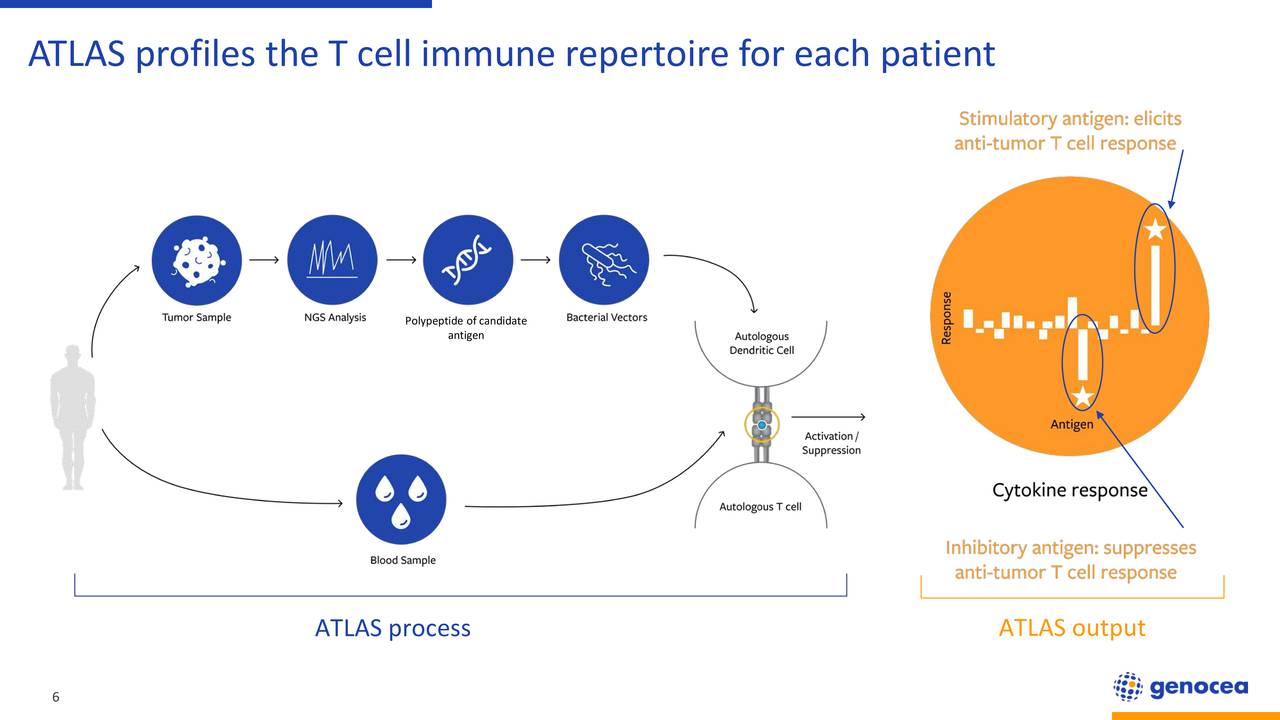
Disease-associated TCR signatures could, thus, be useful biomarkers for immune-mediated disease diagnosis, prognosis, and response to treatment. This robust UMI-based approach has been used successfully to assess the clonal expansion and diversity of the TCR repertoire in peripheral blood of HAM/TSP, multiple sclerosis (MS), and normal healthy donors (NDs), which demonstrated higher clonal T cell expansions in HAM/TSP compared with MS and NDs ( 14). We have recently reported on an unbiased molecular technique that combines the use of unique molecular identifier (UMI), which is barcoded cDNA TCR transcript, with a previously published UMI-based TCR β data sequence analysis pipeline ( 14, 15). Recent technological advances based on high-throughput sequencing (HTS), bioinformatics software, and unbiased gene amplification allow for the analysis of millions of TCR sequences ( 13). Analysis of the TCR repertoire can, thus, provide a better understanding of immune-mediated responses in neuroinflammatory diseases. Importantly, an individual’s TCR repertoire can be altered in the context of infections, malignancies, or immunological disorders. T cells become activated and clonally expanded after they encounter their cognate antigen ( 12). TCR diversity is generated through random rearrangement of variable regions (V), diversity (D) of the β chain, and joining (J) segments of TCR genes (VDJ rearrangements) and is a central component of the adaptive immune system ( 10). Although the actual diversity of human TCR repertoires remains unknown, recent estimates of the potential number of TCR repertoires have been reported to be highly diverse in the range from 1 × 10 13 to 1 × 10 20 in humans ( 9– 11). T cell–mediated antigen recognition depends on the interaction of the T cell receptor (TCR) with antigen–major histocompatibility complex (antigen-MHC) molecules ( 8). It has been suggested that HTLV-1–infected CD4 + T cells and HTLV-1–specific CD8 + cytotoxic T cells enter the CNS and release various proinflammatory cytokines, resulting in the neural damage and degeneration in HAM/TSP patients ( 7). The frequency of these Tax-specific CTLs is extraordinarily high in peripheral blood and even higher in cerebrospinal fluid (CSF) ( 6), in which virus-specific CTLs demonstrate degranulation activity and produce proinflammatory cytokines ( 3, 4). The HTLV-1 regulatory protein Tax promotes the proliferation of HTLV-1–infected lymphocytes and is an immunodominant antigen recognized by HTLV-1–specific CTLs ( 5). 
HTLV-1 predominantly infects CD4 + T cells and causes the activation and proliferation of infected cells, which express viral proteins that may lead to the activation and expansion of HTLV-1–specific CD8 + cytotoxic T lymphocytes (CTLs) ( 3, 4). HAM/TSP is a neuroinflammatory disease of the spinal cord with progressive spastic paraparesis and bladder dysfunction. Human T-lymphotropic type 1 (HTLV-1) infects 10 million–20 million people worldwide and is the etiologic agent of adult T cell leukemia/lymphoma ( 1) and an inflammatory neurologic disease, HTLV-1–associated myelopathy/tropical spastic paraparesis (HAM/TSP) ( 2). These results suggest that exploring TCR repertoires of CSF and antigen-specific T cells may provide a TCR repertoire signature in virus-associated neurologic disorders. Importantly, TCR β clonotypes of expanded clones in HTLV-1 Tax11-19–specific CD8 + T cells were also expanded and enriched in the CSF of the same patient. In addition, we analyzed TCR β repertoires of highly expanded and potentially immunopathologic HTLV-1 Tax11-19–specific CD8 + T cells from PBMCs of HLA-A*0201 + HAM/TSP and identified a conserved motif (PGLAG) in the CDR3 region. The sequence analysis demonstrated that TCR β repertoires in CSF of HAM/TSP patients were highly expanded and contained both TCR clonotypes shared with PBMCs and uniquely enriched within the CSF. 
TCR β libraries using unique molecular identifier–based methodologies were sequenced in paired peripheral blood mononuclear cells (PBMCs) and CSF cells from HAM/TSP patients and normal healthy donors (NDs). In this study, we examined and characterized disease-specific TCR signatures in cerebrospinal fluid (CSF) of patients with HTLV-1–associated myelopathy/tropical spastic paraparesis (HAM/TSP).
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
